“Any one who is not shocked by the quantum theory doesn't understand it.”
Attributed to Niels Bohr by Michio Kaku – Visions, p. 265.
“If you think you understand quantum theory . . . you don’t understand quantum theory.”
Attributed to Richard Feynman by R. Dawkins – The God Delusion,
p.365.
"The story of how people hit on the highly nonintuitive world picture of quantum mechanics, in which the physical state of a system is represented by an element in an abstract linear space and its observable properties by operators in this space, is fascinating and exceedingly complicated.
The theory could not have been deduced from experiment, for the elements of the linear space are in principle not observable. It is also true that the theory did not arise from one person's great insight, as happened in Einstein's discovery of general relativity theory. The much greater change from the classical world picture came in many steps taken by many people, often against the better judgment of participants."
P. J. E. Peebles. 1980. p. 3.
"The quantum theory was was a somewhat bizarre hypothesis that had no connection with classical physics or with any other basic principle. As Max Planck, its originator, was to say later, it was a fortunate guess.
Planck was already well-known by the time he put forward his idea of quanta."
J. Andrade e Silva & G. Lucian (1969) Quanta. p. 60.
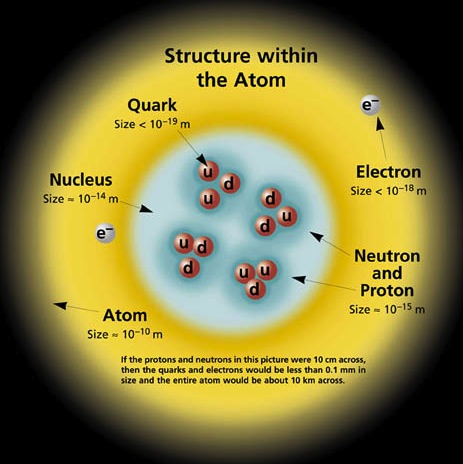
The word quanta from quantum refers to a specified amount indicated by testing that is distinct from and not gradually emergent from another less or more amount. The amount can refer to energy levels in an atom, or an electrical charge, or frequencies, or momentum (velocity).
Related to quantity, originally the quantum referrred to the amount of energy that is proportional to an identified magnitude of the frequency of electromagnetic radiation emitted from atoms in separable intervals.
• Fact •
Slightly more than every ten minutes[1] a stray neutron decays into its binary pair [a proton with positive and an electron with negative charges] as a condition of entropy; when within an atomic nuclei, where neutrons outnumber protons we call this same statistical tendency: beta-decay.

Artist's rendering of beta decay.
We refer to these observed patterns of recurring behavior as predictive.
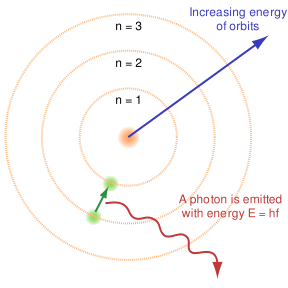
"Light shining on a metal knocks out electrons. Einstein (1905) proposed an interpretation of this effect based on Planck's prescription E = nhw for the energy of an oscillator. Planck's prescription indicates that light can only transfer energy in discreet units – photons, or quanta of the electromagnetic field – of amount how."
P. J. E. Peebles. Quantum Mechanics. 1980. p. 22.
"Quantum Weirdness: It's All in Your Minds,"
Hans Christian von Beer
June, 2013
pp. 47-51
"A new vision of quantum theory sweeps away bizarre paradoxes of the microscopic world."
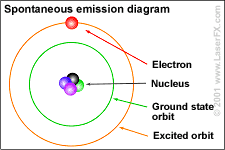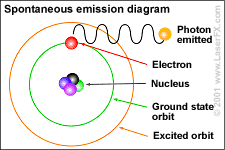
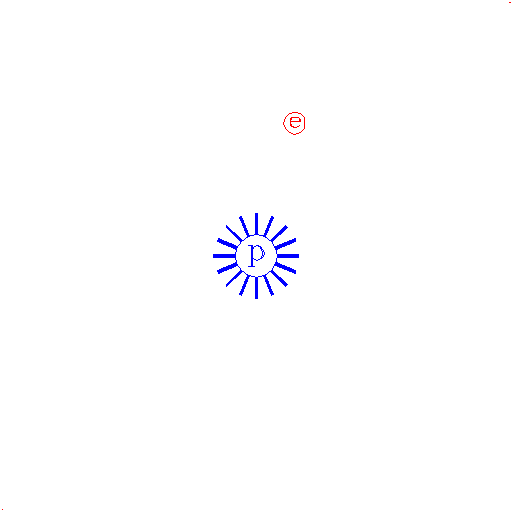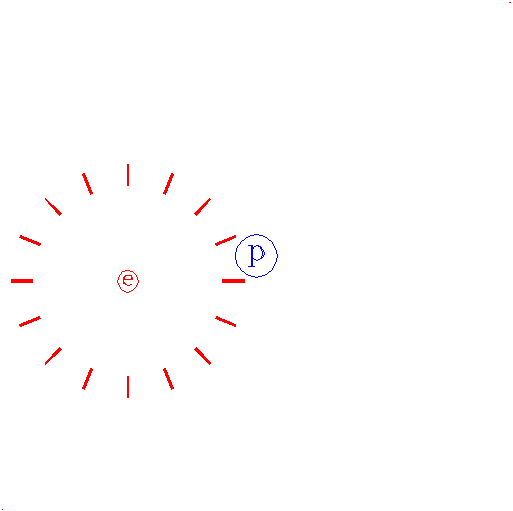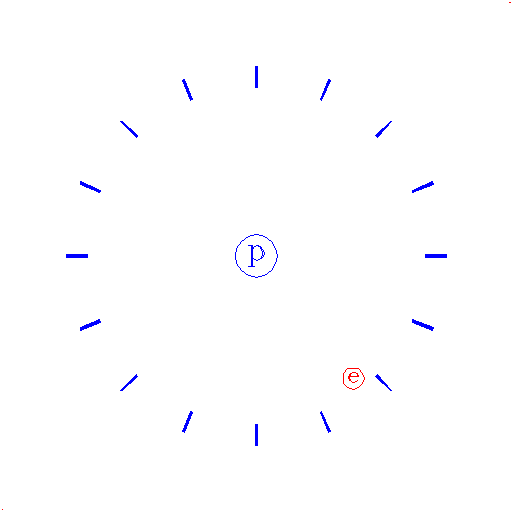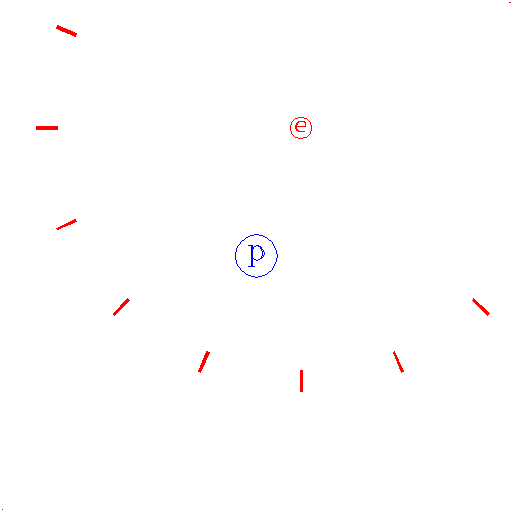
"Consider again the electron."
 |
|
 |
|
The e represents an electron, while the p represents the proton; one electron and one proton by definition is a hydrogen atom.
p. 48.
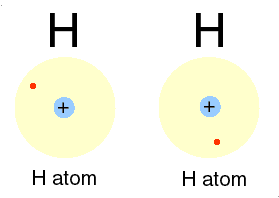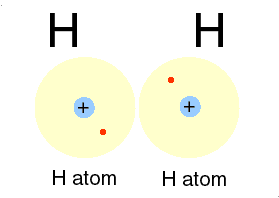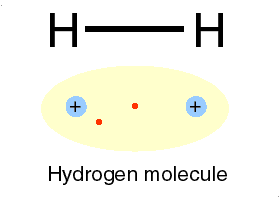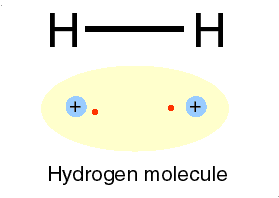

Qbism, [quantum Bayesian] which combines quantum theory with probability (frequentist and Bayesian varieties) theory, maintains that the wave function has no objective reality."
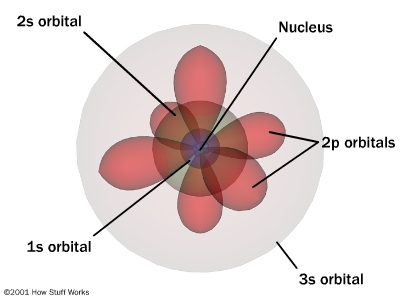
The redescription of the wave function, used by quantum physicists to describe the "movement" of electrons in relation to the nuclei of atoms, is the focus of Qbism.
p. 48.
for more on quantum Bayesian functions, see: "…Paradoxes of Quantum Mechanics".
Science is, in part knowledge of predictable behavior.
How? Any scientist measures repetitive patterns to describe these occurrences so that we understand enough of that behavior to predict the next event.
It is thus one means to know our place in and the character of the material world.
Knowledge of nature means, for example, that:
“All the world is made of the same atoms.”
Feynman, The Meaning of It All: Thoughts of a Citizen-Scientist. (1998), P.12. Se also: http://www.nytimes.com/books/first/f/feynman-meaning.html.
"physics concerns what we can say about nature."
Neil's Bohr,
quoted in "Quantum Weirdness: It's All in Your Minds,"
Hans Christian von Beer Scientific American, June, 2013. pp. 47-51. p. 51.
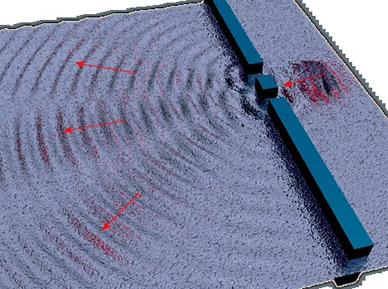
Double Slit Experiment.
"So here is where this comforting prop is knocked away. To begin with, we see that the pattern of fringes on the back of the screen is connected somehow to the way the two waves interfere. Just as with normal waves, its details depend on the width of the slits, the distance between them and how far away the back screen is."
Jim Al-Khalili. Quantum: A Guide for the Perplexed, (2005), pp. 14-15
"The atoms are now behaving like particles, just like the grains of sand. It is as though each atom behaves like a wave when it is confronted by the slits, unless we are spying on it in which case it innocently remains as a tiny particle.
Crazy isn't it?"

J. Al-Khalili Quantum: A Guide for the Perplexed, (2005), pp. 18-19.
"A free neutron will decay with a half-life of about 10.3 minutes but it is stable if combined into a nucleus. The decay of the neutron involves the weak interaction"
http://hyperphysics.phy-astr.gsu.edu/hbase/particles/proton.html
Richard P. Feynman, The Meaning of It All: Thoughts of a Citizen-Scientist. (1998)
J. Andrade e Silva & G. Lucian. Quanta. New York: Mcgraw-Hill,1969.
Jim Al-Khalili. Quantum: A Guide for the Perplexed. London: Weidenfield & Nicolson, 2003.
P. J. E. Peebles. Quantum Mechanics. Princeton. N. J. : Princeton University Press, 1980.
Stephen Hawking. The Universe in a Nutshell. New York: Random House, 2001.
BBC Documentary: Atom 3 The Illusion of Reality "Atom Episode 3 of 3 - The final part of Professor Jim Al-Khalili's documentary series about the basic building block of our universe, the atom. Al-Khalili explores" contemporary quantum mechanics, QED; quantum electrodynamics, Quarks, and Multiverses.
Atoms | Bohr | Electromagnetism | Light quanta | Quantum biology | Quantum scale | Quantum Universe | Radiation is